
 |
 |
 |
1. RADIOACTIVITY
1.1 Natural Radioactivity
Uranium is the heaviest element found in nature, with the atomic number Z = 92. However, the heaviest stable elements found in nature are several isotopes of lead with atomic number Z = 82 (204Pb, 206Pb, 207Pb, and 208Pb found at 1.4%, 24.1%, 22.1%, and 52.1%, respectively) and one isotope of bismuth with atomic number Z = 83 (209Bi). Except for the first natural isotope of lead (204Pb), all of these isotopes are at least partially of radiogenic origin.
All natural elements with atomic numbers Z > 83 are radioactive. These elements decay either by emission of a-particles (energetic helium nuclei), or by emission of negative b-particles (energetic electrons). Both manners of radioactive decay might be followed by emission of g-rays (energetic photons) if the daughter nucleus is formed in an excited state. The a-emission decreases the atomic number Z (number of protons) by 2 and the mass number A (total number of protons and neutrons) by 4. On the other hand, the negative b-emission increases the atomic number Z by 1 (by transforming one neutron in the atomic nucleus into proton), while the mass number A remains constant. As a consequence, we have 4 distinct natural decay series: uranium, actinium, thorium, and neptunium (see Table 1):
1.2 Radioactive Series'
Three of these radioactive decay series' are named after the isotope with the longest half-life. The actinium series is named after one its members, in order to avoid having two series' with identical names. Only three of these decay series' are found in nature: uranium, actinium, and thorium. The isotope 237Np has a half-life much shorter than the geological age of the Earth - about 5 billions of years. Virtually all neptunium decayed within the first 50 millions of years after the Earth was formed.
With regards to depleted uranium, two series' are important: the uranium series (Table 2A) and the actinium series (Table 2B). These tables do not include branches with yields 1% or less [1].
1.3 Calculation of Activity
Activity of a given amount of radioactive material is calculated as the decay constant l (related to the half-life T) multiplied by the number of radioactive nuclei. One kilogram of a pure radioactive isotope with half-life T[sec] has activity R[Bq/kg]:
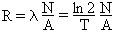
where N = 6.023´1026 /kmol is the Avogadro number, A[kg/kmol] is the mass number, ln 2 = 0.693, and T is the half-life in seconds. For example, A = 235 kg/kmol for 235U and 238 kg/kmol for 238U. The unit of activity is Becquerel (1 Bq = 1 decay/sec) or Curie (1 Ci = 3.7´1010 Bq).
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
1.4 Activity of Uranium Ore
Natural uranium contains 99.274% of 238U, 0.720% of 235U, and 0.0055% of radiogenic 234U [2]. Radioactivity of pure isotopes 238U and 235U is given in Table 4. In uranium ore U3O8 (a crystalline blend of UO2 and UO3 in 1 : 2 ratio, known as pitchblende or "yellow cake"), isotope 238U is in secular equilibrium with 14 other radioactive isotopes, each having the same activity as the isotope 238U, including the radiogenic isotope of uranium 234U. Similarly, isotope 235U is in secular equilibrium with 11 other radioactive isotopes. Therefore, activity of 238U has to be multiplied by a factor of 14´ (compared to the pure 238U) and activity of 235U by a factor of 11´ (compared to pure 235U). If we consider only a-activity, we count only the a-emitters. In this case, activity of 238U has to be multiplied by a factor of 8´ and activity of 235U by a factor of 7´.
1.5 Activity of Natural Uranium
In the process of uranium extraction (all 3 isotopes) from the ore, all radioactive daughter products in both radioactive decay series' are eliminated (with the exception of the radiogenic isotope 234U). A new secular equilibrium is established in a period of a few months between 238U and two other radioactive daughter products (234Th and 234mPa). Secular equilibrium between the uranium isotopes 238U and 234U is not disturbed by the extraction process. Similarly, a new secular equilibrium is established in a period of a few days between the 235U and one other radioactive daughter product, 231Th. Therefore, activity of 238U after the extraction has to be multiplied by a factor of 4´ and activity of 235U by a factor of 2´. If we are concerned only with a-activity, activity of 238U has to be multiplied by a factor of 2´ and activity of 235U remains unchanged.
1.6 Enriched and Depleted Uranium
In order to sustain the chain reaction of nuclear fission, uranium has to be enriched by the fissible isotope 235U to a reactor grade of 3.2 - 3.6% or weapon grade (90%+) uranium. This process not only produces the enriched product, but also a waste stream depleted in 235U, typically to less than 0.3%. The Nuclear Regulatory Commission (NRC) defines depleted uranium (DU) as uranium in which the content of the 235U isotope is less than 0.72%. The military specifications designate that DU used by the Department of Defense (DoD) contains less than 0.3% of 235U. In actuality, DoD uses only DU that contains approximately 0.2% of 235U [33]. In other words, the 235U content in depleted uranium is lowered to 28% of its content in natural uranium.
The DoD claim is supported by mass spectrometry measurements at the Knolls Atomic Power Laboratory [41] leading to 0.202% content of 235U, and also by testing of soil samples at the Aberdeen and Yuma proving grounds [12]. Soil samples with uranium content above the background level by a factor of 100 - 50,000´ carry a DU signature - 0.21% average content of 235U as determined by mass spectrometry [10], [12].
1.7 Activity of Depleted Uranium
The radiogenic isotope 234U is carried away in the enrichment process with at least the same efficiency as the fissible isotope 235U. This is demonstrated by the radioactivity of weapon grade enriched uranium (47 mCi/kg according to [85]). Since the combined a-activity of 90% 235U and 10% 238U is only 1.95 mCi/kg, the minute amount of 234U accounts for the residual a-activity of the enriched material, about 45 mCi/kg. The enrichment factor for 235U is obtained as a ratio of 235U contents in enriched and natural uranium (90 / 0.72 = 125´), while the enrichment factor for 234U is obtained as a ratio of 234U a-activities (45 / 0.335 = 135´). Various sources put the 234U content between 0.0005 - 0.0020% [24], [33], [41], [57], [92] in DU with the 235U content between 0.2 - 0.3%, corresponding to 9 - 36% of the original 234U content in natural uranium. Considering the importance of the 234U content for activity determination of depleted uranium, this number is quite inaccurate.
Since the enrichment of 235U depends solely on the mass number difference DA = 238 - 235 = 3 kg/kmol, a reasonable estimate of the 234U content in depleted uranium can be made from the mass difference DA = 238 - 234 = 4 kg/kmol. If, in a multistage enrichment process, the 235U is depleted to 28% of its original content, 234U should be depleted to 0.28(4/3) = 18% of its original content (see detailed calculation), leaving 0.0010% of 234U in depeleted uranium.
Probably the most accurate measured value of the 234U content in depleted uranium, the one which we use in this review, was obtained by mass spectrometry measurements at the Knolls Atomic Power Laboratory [41]. This value is 0.0008% of 234U in DU with 0.2% of 235U, corresponding to 15% of the original 234U content remaining in depleted uranium. In order to obtain the activity of depleted uranium together with its daughter products, activity of 238U has to be multiplied by a factor of 3.15´ and activity of 235U by a factor 2´ as before. If we are concerned only with a-activity, activity of 238U has to be multiplied by a factor of 1.15´ and activity of 235U remains unchanged. The final results for both the total activity and a-activity of depleted uranium with their daughter products is given in Table 4.
We can assert that the total activity of depleted uranium is 22% less and a-activity 43% less compared to the natural mixture of uranium isotopes, primarily because of the lower content of the radiogenic isotope 234U. This is the origin of the often reported claim that DU is about 40% less radioactive than natural uranium. Depletion of the 235U isotope alone accounts only for 1.6% drop in both the total and a-activity.
Some ridiculous claims have been made recently, comparing radioactivity of depleted uranium to the background radioactivity of lead [84]. For this reason, the background total and a-activity of modern lead is also given in in Table 4. This radioactivity is almost entirely due to the 210Pb contamination, together with its daughter products 210Bi and 210Po (credit to Ettore Fiorini [103], see Table 2A of the uranium decay series). Since the radioactivity of commercial lead is not its intrinsic property (as it is for uranium), it depends on the supplier and the values in Table 4 are typical. Medieval and ancient lead can be several orders of magnitude less radioactive than modern lead (up to 50,000´ less for Roman lead), beause the 210Pb half-life T = 22.3 years is small compared to a few centuries (or millenia) [103]. But the total radioactivity of depleted uranium is still about 60,000´ higher than the total radioactivity of modern lead and its a-activity is 65,000´ higher.
 |
 |
 |
.
.
.
.
.
.
.
.